Abstract
CD47 is a marker of self that provides a "don't-eat-me-signal" through activation of signal-regulatory protein alpha (SIRPa), a cell surface receptor expressed on monocytes/macrophages and granulocytes. This interaction negatively regulates effector functions such as, phagocytosis, migration, and superoxide production. Upregulation of CD47 expression in cancer, including diffuse large B-cell lymphoma (DLBCL), has emerged as a mechanism to escape innate immune surveillance. Using conventional immunohistochemical detection, we assessed CD47 expression in DLBCL and interrogated its association with clinicopathologic features.
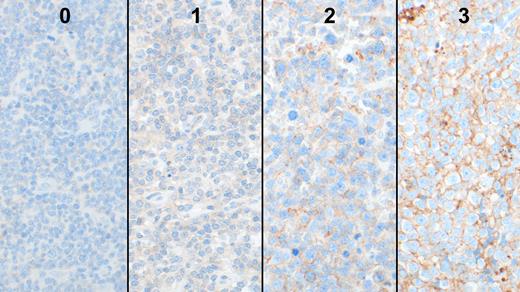
Patients with de novo DLBCL were identified from two large institutions and were uniformly treated with R-CHOP and had sufficient material for study. Immunohistochemical stains (IHC) were performed on FFPE tissue (Hans algorithm, BCL2, MYC, and CD47) and scored semi-quantitatively from no reactivity (0) to strong (2 and 3; Figure 1). Mutational analysis using a 334 gene target sequencing panel, gene expression profiling using Lymph2Cx to determine the cell of origin (COO), and FISH analysis for MYC, BCL2, and BCL6 translocations, were performed. The Lymphgen tool (Wright et al, 2020) was also used to determine the DLBCL group. Fisher's exact test and Kaplan-Meier survival analysis for overall survival (OS) were performed and P <0.05 was considered significant.
CD47 expression was assessed by IHC in a cohort of 152 cases of de novo DLBCL, including 107 cases of germinal center B-cell (GCB) type (70%), 37 cases of activated B-cell (ABC) type (24%), and 8 cases of intermediate type (5%). A total of 17 cases (11%) showed strong and diffuse CD47 expression with IHC scores of 2 or above (CD47hi). CD47hi cases were significantly more frequent in ABC DLBCL (24%, 9/37) than GCB DLBCL (6%, 6/107; P=0.003). The remaining 2 CD47hi cases were in the intermediate DLBCL group (25%, 2/8). ABC DLBCL with CD47hi showed more frequent mutations with TET2 (33% vs 7%; P=0.08) and ZFP36L1 (22% vs 0%; P=0.05) compared to cases with low expression of CD47 with IHC scores of less than 2 (CD47low). ABC DLBCL with CD47low showed more frequent mutations of NOTCH2 (18% vs 0%; P=0.31) and MYD88 (29% vs 11%; P=0.4) compared to CD47hi. GCB DLBCL with CD47hi showed frequent mutations of TP53 (67% vs 21%; P=0.026) and CCND1 (33% vs 0%; P=0.003) compared to CD47low. None of the 13 cases with double- or triple-hit for MYC, BCL2 and/or BCL6 showed CD47 expression. The Lymphgen tool showed that cases of DLBCL with CD47hi were mostly in the 'other' group (50%), with other groups represented such as ST2 (21%), EZB (14%), MCD and BN2 (1 case each). There was no difference in overall survival (OS) between CD47hi and CD47low DLBCL (5-year OS, 75% vs 72%; P=0.57), or with the GCB or ABC subtypes.
Strong expression with CD47 is more frequent in ABC DLBCL and is seen in a subset of GCB DLBCL with mutations in TP53 and/or CCND1. The level of CD47 expression does not appear to predict OS in patients with DLBCL treated with R-CHOP. This study demonstrates that conventional immunohistochemical methods can readily identify DLBCL with high CD47 expression, and these patients may benefit from the use of anti-CD47 therapy.
Herrera: Gilead Sciences: Research Funding; Takeda: Consultancy; Tubulis: Consultancy; Karyopharm: Consultancy; Bristol Myers Squibb: Consultancy, Research Funding; AstraZeneca: Consultancy, Research Funding; ADC Therapeutics: Consultancy, Research Funding; Seagen: Consultancy, Research Funding; Genentech: Consultancy, Research Funding; Merck: Consultancy, Research Funding; Kite, a Gilead Company: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal